GW-0742 – 20MG/ML – 30ML/60ML BOTTLE
$70.99 – $154.98
GW-0742 is sold for laboratory research use only. Terms of sale apply. Not for human consumption, nor medical, veterinary, or household uses. Please familiarize yourself with our Terms & Conditions prior to ordering.
Also Available In:
![]() Powder Option >>
Powder Option >>
![]() Gel Option >>
Gel Option >>
- Description
- Additional information
Description
GW-0742 Liquid
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
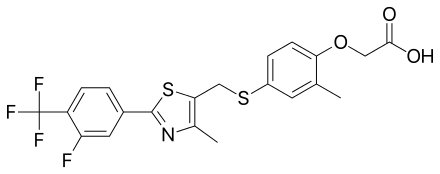
| CAS Number | 317318-84-6 |
| Other Names | GW0742, GW 0742, GW0742X, GW-0742X, 4PZK9FJC4Z, CHEMBL38508, DTXSID9040760, CHEBI:79991, GW 0742X, SR-05000000447, 3tkm |
| IUPAC Name | [4-[[[2-[3-fluoro-4-(trifluoromethyl)phenyl]-4-methyl-5-thiazolyl]methyl]thio]-2-methyl phenoxy]-acetic acid[1] |
| Molecular Formula | C₂₁H₁₇F₄NO₃S₂ |
| Molecular Weight | 471.5 |
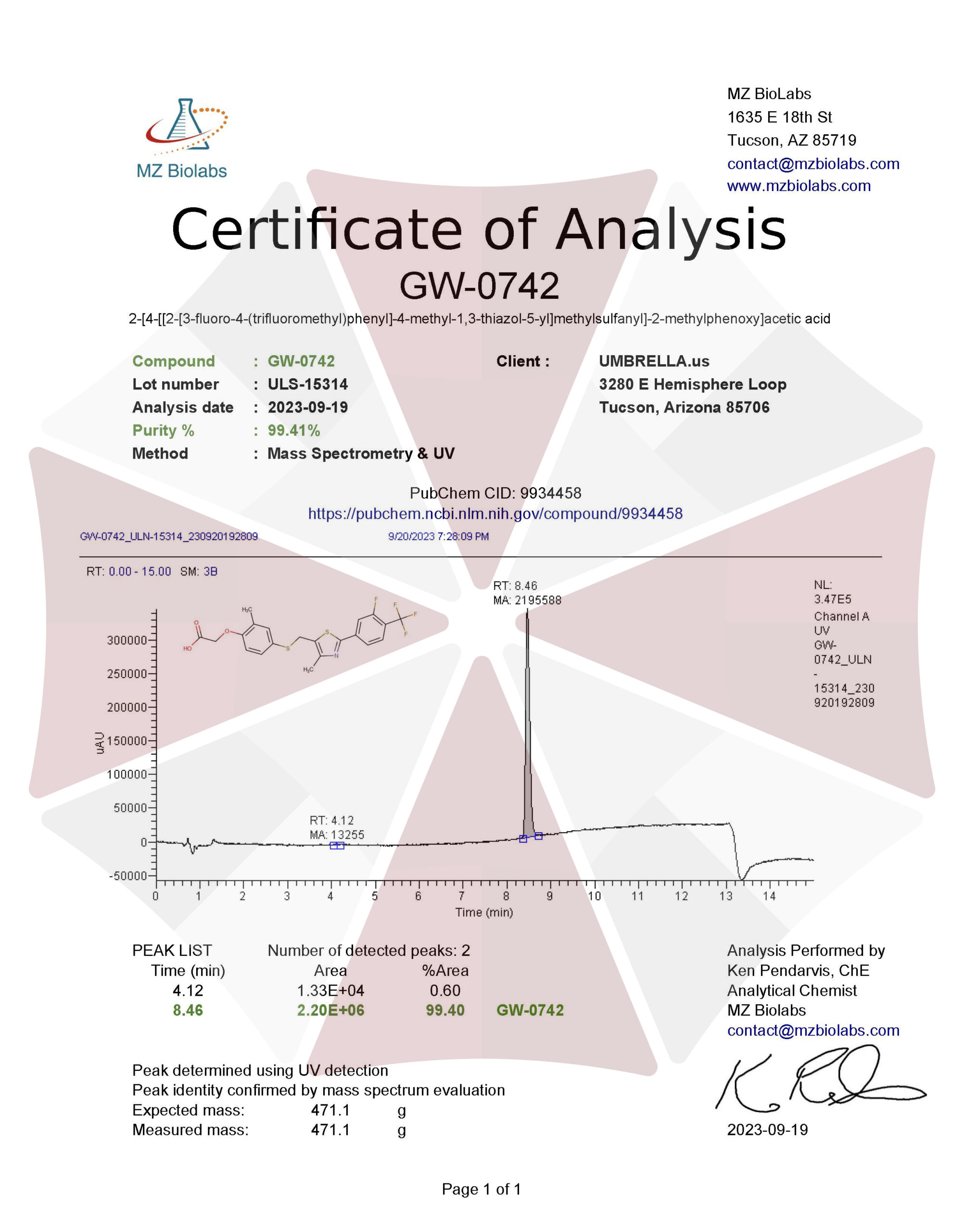
| Purity | ≥99% Pure (LC-MS) |
| Liquid Availability | |
| Powder Availability | |
| Gel Availability | |
| Storage | Store in cool dry environment, away from direct sunlight. |
| Terms | All products are for laboratory developmental research USE ONLY. Products are not for human consumption. |
GW-0742: A next-gen PPAR activator making waves in the SARM research community
Low endogenous testosterone levels frequently develop during anabolic growth cycles, because exogenous androgens such as testosterone esters or SARMs (depending on dose) can be suppressive of endogenous testosterone production in Leydig cells.
A link between low testosterone and diabetes is evident considering that men with type 2 diabetes have lower testosterone levels than weight-matched nondiabetic control subjects [1]. In addition, six large clinical studies have shown that low testosterone levels predict development of insulin resistance and type 2 diabetes in men [2]–[6]. Two additional studies demonstrate a positive relationship between total testosterone levels and insulin sensitivity in normal [7] and diabetic men [8]. Furthermore, it is well established in clinical trials that SARM use can temporarily reduce testosterone levels, albeit in a reversible manner [9]. Taken together, it is clear that there is a need to develop new therapies to prevent insulin resistance which may develop as a consequence of therapeutic SARM use.
The development of new drugs for the treatment of insulin resistance is critically important, not only for patients with diabetes but for patients who develop insulin resistance as a consequence of anabolic supplement use, including SARMs. GW-0742, a selective peroxisome proliferator-activated receptor (PPAR)-δ activator, has already been shown to prevent metabolic abnormalities including insulin resistance in skeletal muscle.
Main Pre-Clinical Research Findings for GW-0742:
- Enhances hepatic (liver) fat metabolism [10]
- Reduces liver stress by improving hepatic energy metabolism [11]
- Prevents hypertension and vascular inflammation [12]
- Demonstrates anti-inflammatory effects in lungs [13]
- Increases cardiac vascularization [14]
- Prevents neuron cell death following electrolyte imbalance [15]
Selected Preclinical Studies:
- “GW0742 (PPAR-beta activator) attenuates hepatic endoplasmic reticulum stress by improving hepatic energy metabolism in high-fat diet fed mice” [11]
Liver stress and hepatic steatosis are intertwined with insulin resistance. PPARs are at the crossroads of these pathways. This study aimed to investigate the effects of GW0742 (PPAR-beta activator) on hepatic energy metabolism and liver stress in a murine diet-induced obesity model. High-fat diets cause individuals to become overweight, show hyperinsulinemia (ie. high insulin levels), hepatic inflammation and hepatic lipogenesis (ie. synthesis of new liver fat), leading to overall liver stress, with structural and molecular alterations, ultimately resulting in cellular death. However, GW0742 prevented increases in body weight and improved glucose tolerance, tackled hepatic inflammation and favored hepatic metabolism versus lipogenesis, thus leading to less accumulation of liver fat. These results are consistent with structural improvements, reducing liver stress and liver cell death in treated individuals. These results indicate that the PPAR-beta/delta activation alleviated liver stress by improving insulin sensitivity and maximizing the hepatic energy metabolism. PPAR-beta/delta activation by GW0742 could be an essential tool to avoid fatty-liver progression and other metabolic disorders.
Currently, obesity is considered a worldwide epidemic, affecting different age groups in both developed and underdeveloped countries (Aasheim & Sovik, 2011). Obesity is a hallmark of insulin resistance (IR) and inflammation development, favoring the type 2 diabetes mellitus onset (Seidell & Halberstadt, 2015). Obesity pathogenesis is related to surplus energy intake coupled with reduced energy expenditure (Mokdad et al., 2003). In this regard, excessive saturated fatty acids intake disrupts many pathways, altering the balance among different PPAR isoforms in the liver, turning it more prone to inflammation and glucolipotoxicity (Valenzuela et al., 2015).
Inflammation and insulin resistance frequently affect the liver, a central organ that is crucial to the regulation of energy metabolism (Gustafson & Smith, 2015). Insulin resistance induces high rates of lipolysis in the white adipose tissue (fatty tissue), reducing its capacity to store lipids and increasing the delivery of fatty acids to the liver (De Minicis et al., 2013; Fan & Cao, 2013). Moreover, insulin resistance impairs hepatic mitochondrial energy production and reduces the hepatic export of lipoproteins. As a result, lipid droplets are deposited within the hepatic tissue, leading to non-alcoholic fatty liver disease (NAFLD) (Angulo, 2014).
The pathogenesis of fatty-liver disease and liver stress are closely intertwined with insulin resistance, obesity and inflammation, showing a rising prevalence worldwide (Hotamisligil, 2010; Saponaro et al., 2015). Liver stress is characterized by the formation of poorly folded and unfolded proteins, producing an imbalance between the cellular demand for folding and maturation of proteins, which facilitates the accumulation of unfolded proteins in central cells of the liver (Yoshida, 2007). In this context, the activation of PPARs by novel therapeutics like GW0742 is emerging as a relevant tool to manage liver stress by modulating hepatic energy metabolism and several altered metabolic routes usually disrupted in obesity, but these effects remain to be elucidated (Schuppan & Schattenberg, 2013). This present study was designed to investigate the effects of the treatment with the PPAR-beta/delta agonist GW0742 on hepatic energy metabolism and liver stress in a diet-induced obesity model.
Study Conclusion:
This study demonstrates that treatment with GW0742 reduced body mass, without altering the energy intake, and alleviated the liver stress by improving insulin sensitivity and maximizing hepatic energy metabolism with a shift towards beta-oxidation instead of lipogenesis (ie. fat accumulation). It can be argued that PPAR modulation is an essential mediator of these present findings and PPAR-beta activation seems to be decisive to restore liver homeostasis, with structural liver cell enhancement. This study has major translational potential as PPAR-beta activation could be a promising tool to prevent fatty-liver progression and other metabolic disorders.
- “Chronic peroxisome proliferator-activated receptorβ/δ agonist GW0742 prevents hypertension, vascular inflammatory and oxidative status, and endothelial dysfunction in diet-induced obesity” [12]
Vascular dysfunction plays a key role in obesity-induced risk of cardiovascular disease. The aim of this study was to analyze the effect of chronic PPAR β/δ activator GW-0742 treatment on vascular function in subjects fed a high-fat diet (HFD).
Obesity is a complex chronic disease whose prevalence is rising. Consequently, the prevalence of obesity-related disorders, such as diabetes and cardiovascular diseases, is also rising. The mechanisms by which obesity increases the risk of cardiovascular disease are incompletely understood, but vascular dysfunction seems to play a key role that can subsequently promote the development of atherosclerosis and hypertension. Many factors may impair vascular function in obesity, including changes in blood pressure, glucose levels, insulin sensitivity and fat metabolism [16]. Further-more, obesity is characterized by a chronic immune-mediated low-grade systemic inflammation [17]. Oxidative stress has also been implicated in vascular dysfunction.
In this study, researchers wanted to test how GW-0742 could modulate hypertension and markers of vascular health in the context of diet-induced obesity. The role of PPAR in obesity has been examined by the loss-of-function approach or by using synthetic PPAR activators. PPAR knockout animals are more prone to weight gain on a high-fat diet (HFD), whereas transgenic animals that have overactivated PPAR are protected against obesity and fat accumulation [18]. Moreover, PPAR activators can improve blood-lipid profiles, increasing the levels of good cholesterol while simultaneously decreasing bad cholesterol and triglycerides in obese animals [19] and overweight men [20]. In addition, PPAR has already been shown to regulate glucose levels and insulin sensitivity in various organs and tissues [21].
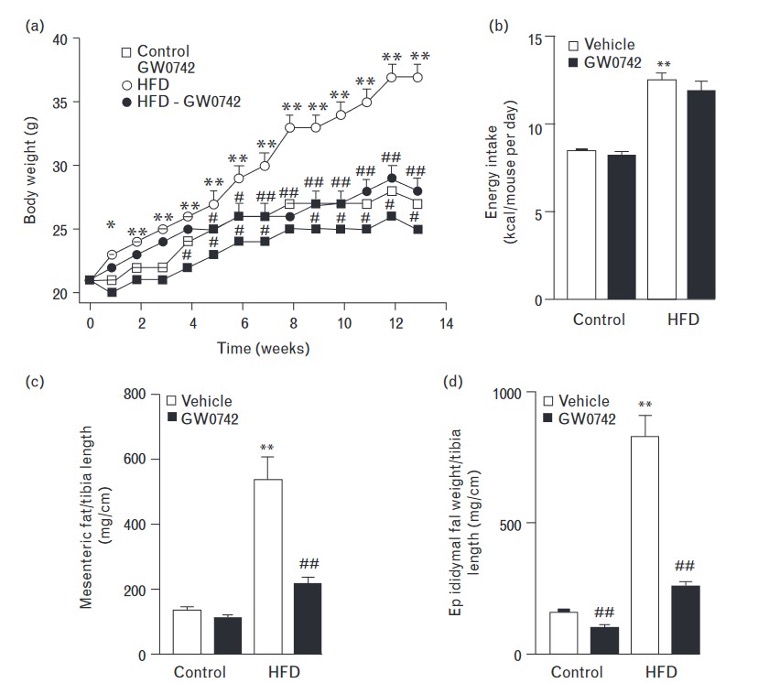
Fig.1. Effects of GW0742 administration on body weight evolution (a), energy intake (b), mesenteric fat (c), and epididymal fat (c) in control and HFD-fed anabolic animals. HFD=High Fat Diet. Control = regular diet.
Study Conclusion
GW0742 prevents obesity, hypertension, vascular dysfunction, and the vascular pro-oxidant and pro-inflammatory state in high-fat diet subjects during an anabolic growth cycle.
Overall Conclusions
GW-0742 is a promising candidate to enhance liver metabolism and reduce liver stress, act as an anti-inflammatory agent, normalize insulin sensitivity, and reduce neuronal stress. Given the relationship between anabolic supplement use and metabolic disturbances, GW-0742 is an exciting new lead to potentially maximize the benefits of SARMs while simultaneously mitigating their risk profiles.
This information is for educational purposes only and does not constitute medical advice. THE PRODUCTS DESCRIBED HEREIN ARE FOR RESEARCH USE ONLY. All clinical research must be conducted with oversight from the appropriate Institutional Review Board (IRB). All preclinical research must be conducted with oversight from the appropriate Institutional Animal Care and Use Committee (IACUC) following the guidelines of the Animal Welfare Act (AWA).
References
[1] E. Barrett-Connor, “Lower endogenous androgen levels and dyslipidemia in men with non-insulin- dependent diabetes mellitus,” Ann. Intern. Med., 1992.
[2] G. Tibblin, A. Adlerberth, G. Lindstedt, and P. Björntorp, “The pituitary-gonadal axis and health in elderly men: A study of men born in 1913,” Diabetes, 1996.
[3] S. M. Haffner, J. Shaten, M. P. Stern, G. D. Smith, and L. Kuller, “Low levels of sex hormone-binding globulin and testosterone predict the development of non-insulin-dependent diabetes mellitus in men,” Am. J. Epidemiol., 1996.
[4] R. K. Stellato, H. A. Feldman, O. Hamdy, E. S. Horton, and J. B. Mckinlay, “Testosterone, sex hormone-binding globulin, and the development of type 2 diabetes in middle-aged men: Prospective results from the Massachusetts Male Aging Study,” Diabetes Care, 2000.
[5] J. Y. Oh, E. Barrett-Connor, N. M. Wedick, and D. L. Wingard, “Endogenous sex hormones and the development of type 2 diabetes in older men and women: The Rancho Bernardo Study,” Diabetes Care, 2002.
[6] D. E. Laaksonen et al., “Testosterone and Sex Hormone-Binding Globulin Predict the Metabolic Syndrome and Diabetes in Middle-Aged Men,” Diabetes Care, 2004.
[7] E. B. Geer and W. Shen, “Gender differences in insulin resistance, body composition, and energy balance,” Gend. Med., 2009.
[8] K. I. Birkeland, K. F. Hanssen, P. A. Torjesen, and S. Vaaler, “Level of sex hormone-binding globulin is positively correlated with insulin sensitivity in men with type 2 diabetes,” J. Clin. Endocrinol. Metab., 1993.
[9] J. T. Dalton et al., “The selective androgen receptor modulator GTx-024 (enobosarm) improves lean body mass and physical function in healthy elderly men and postmenopausal women: Results of a double-blind, placebo-controlled phase II trial,” J. Cachexia. Sarcopenia Muscle, vol. 2, no. 3, pp. 153–161, 2011.
[10] S. C. Kuo, P. M. Ku, L. J. Chen, H. S. Niu, and J. T. Cheng, “Activation of receptors δ (PPARδ) by agonist (GW0742) may enhance lipid metabolism in heart both in vivo and in vitro,” Horm. Metab. Res., 2013.
[11] F. M. Silva-Veiga, T. L. Rachid, L. de Oliveira, F. Graus-Nunes, C. A. Mandarim-de-Lacerda, and V. Souza-Mello, “GW0742 (PPAR-beta agonist) attenuates hepatic endoplasmic reticulum stress by improving hepatic energy metabolism in high-fat diet fed mice,” Mol. Cell. Endocrinol., 2018.
[12] M. Toral et al., “Chronic peroxisome proliferator-activated receptorβ/δ agonist GW0742 prevents hypertension, vascular inflammatory and oxidative status, and endothelial dysfunction in diet-induced obesity,” J. Hypertens., 2015.
[13] Z. Haskova et al., “Modulation of LPS-induced pulmonary neutrophil infiltration and cytokine production by the selective PPARβ/δ ligand GW0742,” Inflamm. Res., 2008.
[14] N. Wagner et al., “Peroxisome proliferator-activated receptor β stimulation induces rapid cardiac growth and angiogenesis via direct activation of calcineurin,” Cardiovasc. Res., 2009.
[15] S. A. Smith, G. R. Monteith, J. A. Robinson, N. G. Venkata, F. J. May, and S. J. Roberts-Thomson, “Effect of the peroxisome proliferator-activated receptor β activator GW0742 in rat cultured cerebellar granule neurons,” J. Neurosci. Res., 2004.
[16] A. Avogaro and S. V. De Kreutzenberg, “Mechanisms of endothelial dysfunction in obesity,” Clinica Chimica Acta. 2005.
[17] G. S. Hotamisligil, “Inflammation and metabolic disorders,” Nature. 2006.
[18] Y. X. Wang et al., “Peroxisome-proliferator-activated receptor δ activates fat metabolism to prevent obesity,” Cell, 2003.
[19] W. R. Oliver et al., “A selective peroxisome proliferator-activated receptor δ agonist promotes reverse cholesterol transport,” Proc. Natl. Acad. Sci. U. S. A., 2001.
[20] U. Risérus et al., “Activation of peroxisome proliferator-activated receptor (PPAR)δ promotes reversal of multiple metabolic abnormalities, Reduces oxidative stress, and increases fatty acid oxidation in moderately obese men,” Diabetes, 2008.
[21] C. H. Lee et al., “PPARδ regulates glucose metabolism and insulin sensitivity,” Proc. Natl. Acad. Sci. U. S. A., 2006.
GW-0742 is sold for laboratory research use only. Terms of sale apply. Not for human consumption, nor medical, veterinary, or household uses. Please familiarize yourself with our Terms & Conditions prior to ordering.











| File Name | View/Download |
| 09-19-2023-Umbrella-Labs-GW-0742-Certificate-Of-Analysis-COA.pdf |
VIEW CERTIFICATES OF ANALYSIS (COA)
Additional information
| Weight | 2 oz |
|---|---|
| Dimensions | 3 × 3 × 5 in |
| Formula Option | |
| CAS Number | |
| PubChem CID | |
| ChEMBL ID | |
| ChemSpider ID | |
| Molecular Formula | C21H17F4NO3S2 |
| Molar Mass | 471.5 g·mol−1 |
| Size |